Hemgenix given to 1st patient in Florida at Tampa General Hospital
Third hospital in US to successfully administer hemophilia B gene therapy
Written by |
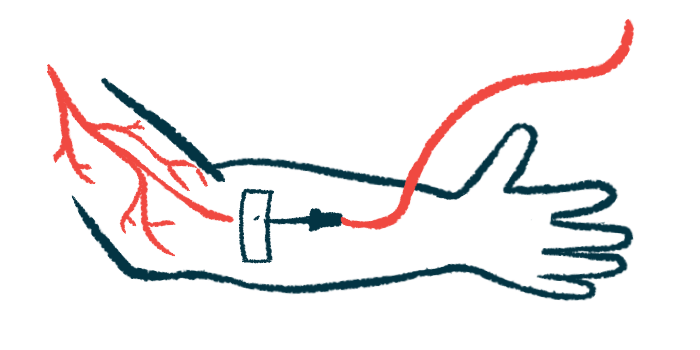
Tampa General Hospital’s cancer institute is the first healthcare center in Florida, and the third in the U.S., to successfully administer the first and only hemophilia B gene therapy, Hemgenix (etranacogene dezaparvovec), to a patient.
“The infusion of this therapy is a hallmark of the TGH Cancer Institute’s dedication to propelling innovation to improve outcomes and quality of life in a meaningful way for our hematology/oncology patients,” Eduardo M. Sotomayor, MD, vice president and executive director of the institute, said in a press release.
“We are proud that in offering this cutting-edge genetic therapy in our newly built, state-of-the-art infusion centers, we are uniquely positioned to address unmet needs for patients living with hemophilia B in our community and beyond,” Sotomayor said.
Hemophilia is characterized by the lack or dysfunction of certain clotting factors, or blood-clotting proteins, and is marked by excessive and prolonged bleeding. Hemophilia B, the second most common type of the condition, is caused by mutations in the F9 gene, which gives instructions for making clotting factor IX (FIX). Because the F9 gene is on the sex-determining X chromosome, hemophilia B is much more common in men than in women.
Hemgenix designed to treat severe to moderately severe hemophilia B
Those with severe or moderately severe hemophilia B, which Hemgenix is designed to treat, have FIX activity levels that are 2% or lower than normal. This puts patients at a high risk of experiencing excessive and protracted bleeds.
The one-time infusion therapy delivers to liver cells a healthy copy of the gene encoding FIX. In doing so, the gene therapy is expected to restore the body’s ability to make functional FIX. Consequently, clotting activity normalizes and the need for FIX replacement treatments is mitigated or eliminated.
“This is an exciting opportunity to pioneer this treatment option, which will allow us to continue expanding access to a wide range of therapies for patients with hemophilia in our community and throughout the state,” said Nathan Visweshwar, MD, Tampa General Hospital’s hematology and oncology specialist who directed the Hemgenix infusion.
The gene therapy was approved in the U.S. in 2022, followed by Canada last year, and conditionally approved in the European Union and the U.K. This month, it was approved in Switzerland for men, 18 and older, with severe or moderately severe hemophilia B.
Hemgenix approvals based on data from Phase 3 HOPE-B trial
Hemgenix approvals were based largely on data from a Phase 3 clinical trial called HOPE-B (NCT03569891), which is evaluating the outcomes of more than 50 men with moderate to severe hemophilia B who were given a single dose of Hemgenix.
Top-line trial results showed Hemgenix increased FIX activity as intended, with the latest data revealing that after three years, FIX activity levels remained heightened at 38.6% of normal, on average.
HOPE-B findings also indicated Hemgenix was safe overall, and that the therapy helped lower bleed rates and the need for conventional replacement treatments.
“Our team’s successful administration of this gene therapy is the perfect case study for the distinct benefits an academic and research health system can deliver to patients,” said Abraham Schwarzberg, MD, a senior vice president and chief of oncology at Tampa General. “The introduction of a new therapeutic option for this rare disease is a testament to our shared commitment to apply the latest research and clinical best practices to safely and effectively treat even the most complex conditions.”




Leave a comment
Fill in the required fields to post. Your email address will not be published.