Advate (octocog alfa) for hemophilia
What is Advate for hemophilia?
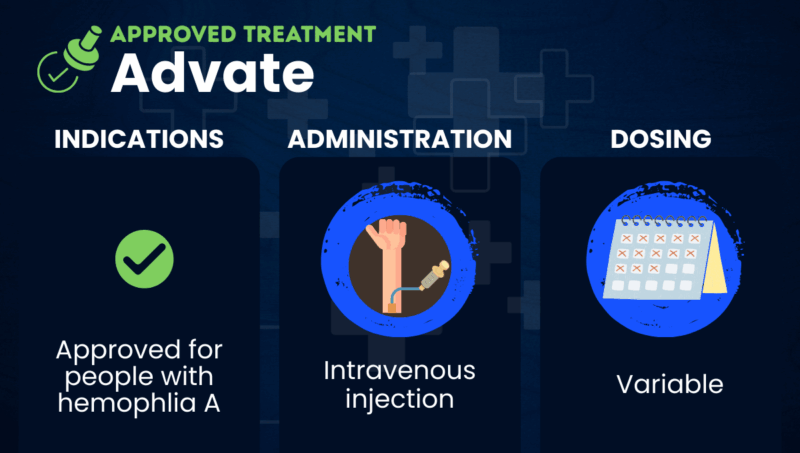
Advate (octocog alfa) is a lab-made clotting factor VIII (FVIII) concentrate approved in the U.S. to prevent and control bleeding in children and adults with hemophilia A. The therapy is indicated for preventive (prophylactic) and on-demand use, as well as to manage bleeding during surgery.
People with hemophilia A have faulty or missing versions of the protein FVIII, which helps blood form clots to stop bleeding. As a result, they may experience heavy, prolonged bleeding after an injury or surgery. In more severe cases, bleeding can occur spontaneously with no clear cause.
Advate, a replacement therapy, contains a recombinant, or lab-made, protein called octocog alfa, which closely resembles FVIII. This can temporarily replace the faulty or missing FVIII, helping prevent and control bleeding. The protein is produced using genetically engineered hamster ovary cells.
Advate is given by intravenous (into-the-vein) injection. It was developed by Baxter and its spinoff, Baxalta, now part of Takeda Pharmaceuticals. Takeda also markets Adynovate (rurioctocog alfa pegol), a modified version of Advate that’s designed to last longer in the body.
Therapy snapshot
| Brand name | Advate |
| Chemical name | Octocog alfa |
| Usage | Prophylaxis and on-demand treatment of bleeding in hemophilia A |
| Administration | Intravenous injection |
Who can take Advate?
In the U.S., Advate is approved for children and adults with hemophilia A. It has three main indications:
- on-demand: to control active bleeding episodes
- prophylactic: to prevent or reduce the frequency of bleeds
- perioperative: to manage the risk of bleeding before, during, and after surgical procedures
Advate is similarly approved in other areas, including Canada and the European Union.
The therapy is contraindicated, or should not be used, in people with a known history of life-threatening reactions to mouse or hamster proteins or to any other ingredients in Advate.
It is also not indicated for people with von Willebrand disease, another type of bleeding disorder.
How is Advate administered?
Advate is delivered as an injection directly into the bloodstream lasting no more than five minutes. The dosage depends on several factors, including the location and extent of the bleeding, the patient’s body weight, and the severity of their FVIII deficiency.
A healthcare provider will inform patients exactly how and when the medication should be administered for on-demand and/or prophylactic use.
On-demand treatment is usually administered at regular intervals until bleeding resolves. When used prophylactically, infusions may be administered once every two or three days, with adjustments based on clinical response.
Some patients can self-administer Advate at home using a prepackaged infusion device, but only after training and approval from a healthcare provider.
Advate in clinical trials
Results from several clinical trials involving people with hemophilia A supported the U.S. approval of Advate.
- A Phase 2/3 clinical trial, Study 069901, demonstrated that Advate effectively controlled and prevented bleeds when used prophylactically and on demand in previously treated patients ages 10 and older. The therapy worked as well as Recombinate, another FVIII replacement therapy from Takeda that is now discontinued.
- A subsequent continuation study (NCT00157053) showed that Advate continued to control the majority of bleeding episodes effectively.
- Another Phase 2/3 clinical trial (NCT00157105) tested the use of Advate to manage surgery-related bleeding among patients ages 5 and older. Bleed control both during and after the procedures was rated as excellent or good in over 90% of cases.
- A Phase 4 trial (NCT00243386) demonstrated that switching from on-demand to preventive use of Advate significantly reduced the number of bleeding episodes in previously treated adults and children 7 years and older.
- A Phase 3 pediatric study (NCT00157157) showed that most bleeding episodes could be effectively managed with one or two Advate infusions in children younger than 6 who had received no or minimal previous treatment. Another study also demonstrated good bleed control for previously treated pediatric patients.
Advate side effects
The most common side effects associated with Advate are:
- fever
- headache
- cough
- the common cold
- joint pain
- vomiting
- upper respiratory tract infection
- limb injury
- nasal congestion
- diarrhea
An uncommon, but potentially serious side effect is allergic reactions, including systemic reactions (anaphylaxis) that can be life-threatening. Some people may develop a sensitivity to mouse or hamster protein, which is present in trace amounts.
Signs of a hypersensitivity reaction may include dizziness, numbness, tingling, or prickling sensations, rash, flushing, facial swelling, hives, shortness of breath, itching, or vomiting. Should symptoms of hypersensitivity occur, Advate should be discontinued.
Some people may develop inhibitor antibodies that neutralize FVIII and may stop Advate from working properly. Patients will be monitored for the development of inhibitors.
Hemophilia News Today is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by